Drug Cases -
Zantac and Singulair Cases
Beck Law Center is a pioneer in pharmaceutical cases. Kim Beck filed some of the earliest Zantac Cases. As of Mid-September 2021, Beck Law Center had the only filed Singulair cases. By early 2022, Beck Law Center started working will every firm that started filing cases.
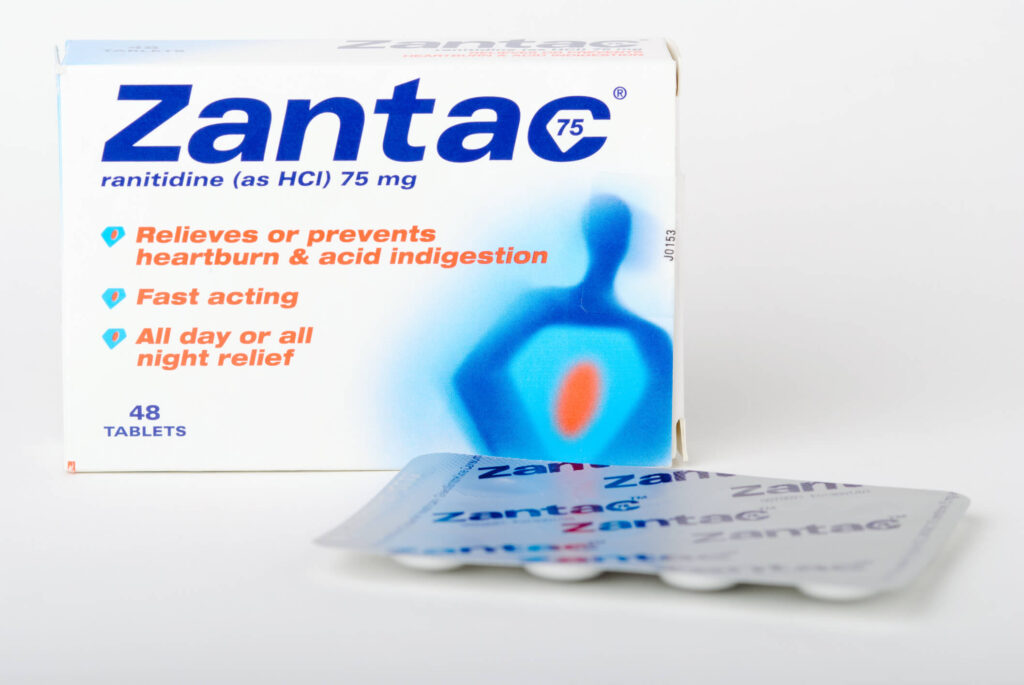
Zantac
Zantac and its active ingredient ranitidine have been sold in the United States since 1983, originally only as a prescription, and later also as an over the counter treatment for acid indigestion, heartburn, and sour stomach. Tens of thousands of people used Zantac every year, not knowing the extent of its danger.
Singulair
Singulair has been on the market since 1998. It is "indicated for" (meaning it is supposed to treat) asthma, hay fever and allergies. From 1998 on, Merck has received reports of neuropsychiatric injuries, like OCD, depression, tics, tremors, and suidicality. Regardless, Merck has played down Singulair's risks.
Finally in March of 2020, the FDA forced Merck to add a "Black Box Warning" to Singulair's label. This is the strongest kind of warning the FDA can require.
Let's talk about your case.
You can contact us at information@becklawcenter.com, 888-434-2912, or by filling out the form below. You can also click Book an Appointment with a Lawyer Online to schedule a free consultation.
Click here for our mailing address and other contact information.
Thanks for your message! We’ll be in touch soon.
